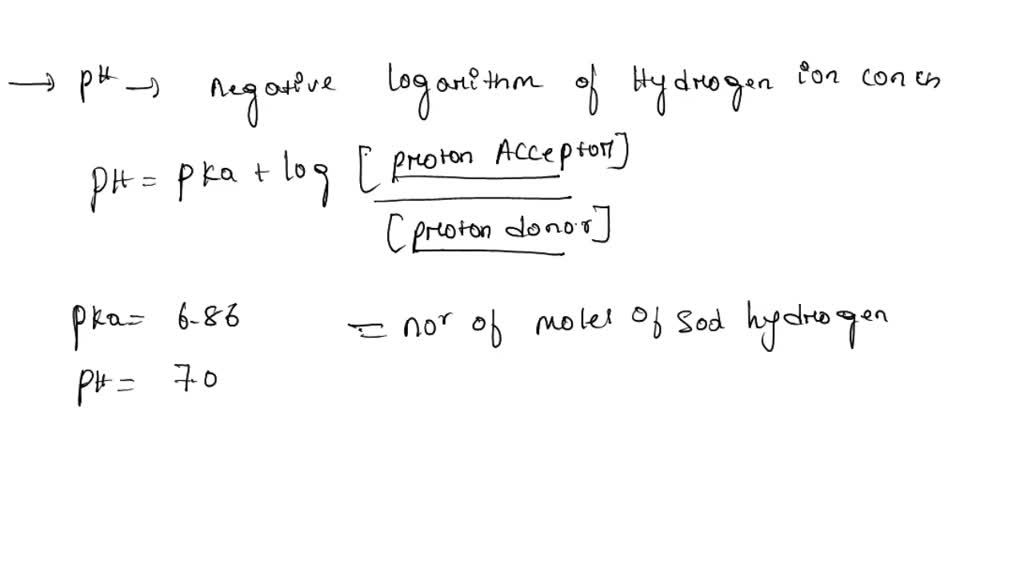
SOLVED: Problem for the group: Determine the weight in grams of sodium dihydrogen phosphate (NaH2PO4.H2O; FW = 138.01) and sodium hydrogen phosphate (Na2HPO4; FW = 141.98) needed to prepare 1L of a

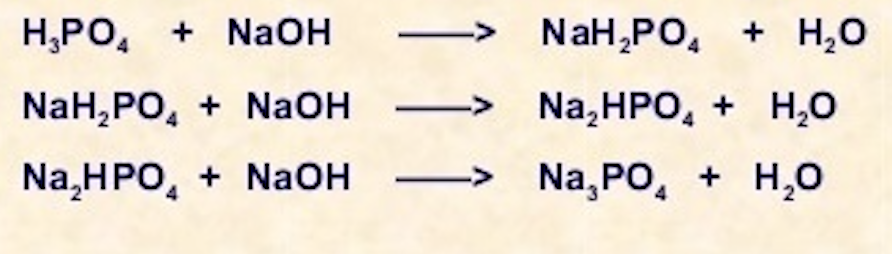
ReasonH_3PO_4 is a tribasic acid.AssertionNaOH + H_3PO_4 rightarrow NaH_2PO_4 + H_2O In the given reaction, the equivalent weight of H_{3}PO_{4} is M/3.
![Sodium Phosphate Monobasic dihydrate (NaH2PO4.2H2O, 1kg) [CN04-1KG] - $40.00 : Bioland Scientific, for Your Research Needs Sodium Phosphate Monobasic dihydrate (NaH2PO4.2H2O, 1kg) [CN04-1KG] - $40.00 : Bioland Scientific, for Your Research Needs](https://www.bioland-sci.com/images/NaH2PO4s%201KG.jpg)
Sodium Phosphate Monobasic dihydrate (NaH2PO4.2H2O, 1kg) [CN04-1KG] - $40.00 : Bioland Scientific, for Your Research Needs

SOLVED: A buffer was made by mixing aqueous solutions of NaH2PO4 and Na2HPO4 together. This buffer is made by mixing two salts together. a. Write the balanced dissociation reaction for solid NaH2PO4
8. Calculate equivalent weight of H3PO4 and Ca(OH)2 on the basis of given reaction H3PO4 + NaOH = NaH2PO4 + H2O Ca(OH)2 + HCl = Ca(OH)Cl + H2O
Sodium Dihydrogen Phosphate Starting From Sodium Chloride and Orthophosphoric Acid Via Cation Resin Exchange

NaOH+H3PO4=NaH2PO4+H2O Balanced Equation||Sodium hydroxide+Phosphoric acid Balanced Equation - YouTube

Mono Sodium Phosphate Water Treatment Chemicals Nah2po4. H2O - China Monosodium Phosphate, Sodium Dihydrogen Phosphate | Made-in-China.com
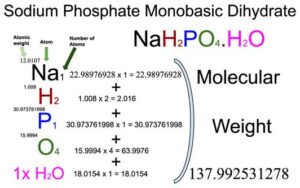
Sodium Phosphate Monobasic Monohydrate (NaH2PO4.H2O) Molecular Weight Calculation - Laboratory Notes
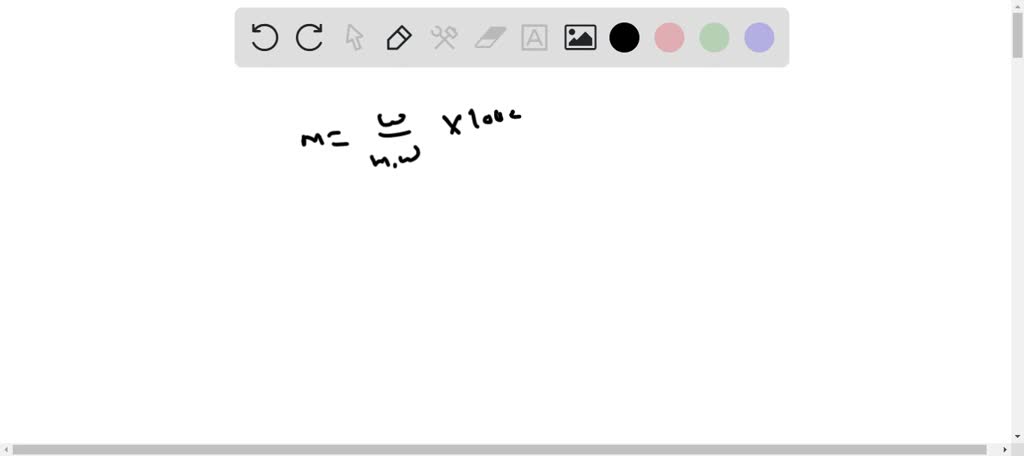
SOLVED: Weigh out 10.4 g sodium phosphate (NaH2PO4.H2O, MW = 138 g/mol). Dissolve the NaH2PO4.H2O in 450 mL water. Bring pH of solution to 7.4 with 10 M NaOH (need about 2










.jpg)