a) Sodium thiosulfate rod raw material (Na2S2O3 · 5H2O), a spherical... | Download Scientific Diagram
Natrium Tiosulfat Pentahidrat | Na2S2O3.5H2O | Sodium Thiosulfate Pentahydrate | 500 g/botol | Lazada Indonesia
The amount of Na2S2O3.5H2O required to completely reduce 100 mL of 0.25 N iodine solution, is (A) 6.20 g(B) 9.30 g(C) 3.10 g(D) 7.75 g

1.48 Sodium thiosulphate (Na, S,O3 .5H2O) reacts with iodine as follows: 25203 + 12 -$40% + 21 Calculate the equivalent mass of Na2S2O3.
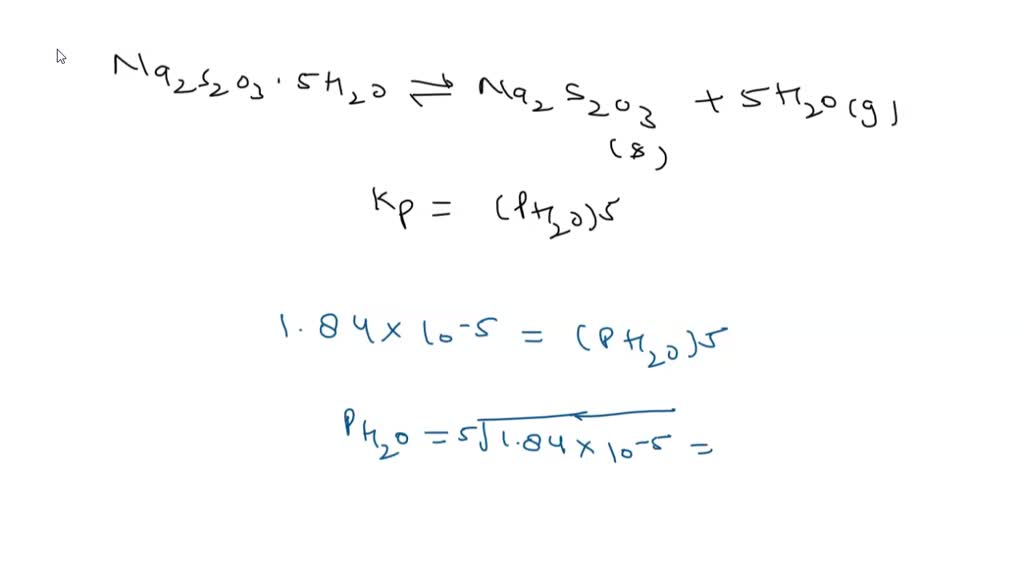
SOLVED: Sodium thiosulfate pentahydrate (FM 248.18 g ∙ mol-1) loses water when it is heated in an oven: Na2S2O3∙5H2O(s) ⇌ Na2S2O3(s) + 5H2O(g). ΔH° and ΔS° for this reaction at 25°C are

Le thiosulfate de sodium, hyposulfite de sodium, Na2S2O3 5H2O - Chine Hyposulfite de sodium, de thiosulfate de sodium

Na2S2O3 5H2O du thiosulfate de sodium Pharma Grade, cristaux incolores - Chine Le Sodium, de thiosulfate de sodium Thiosulfate de sodium

SOLVED: The compound sodium thiosulfate pentahydrate, Na2S2O3·5H2O, is important commercially to the photography business as "hypo", because it has the ability to dissolve unreacted silver salts from photographic film during development. Sodium