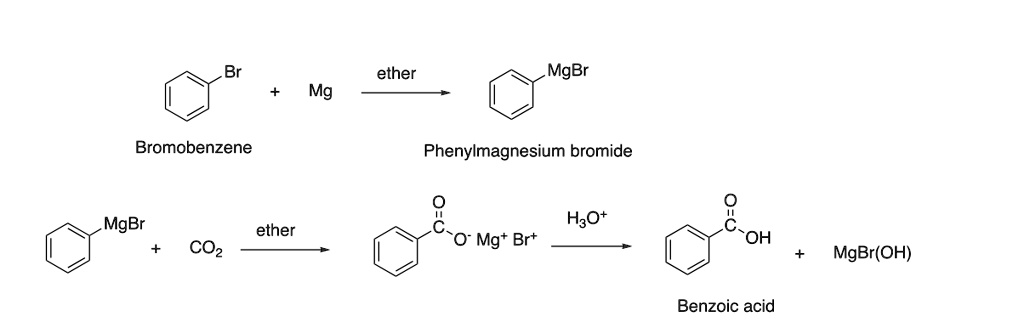
SOLVED: Ether MgBr Mg Bromobenzene Phenylmagnesium bromide MgBr ether H2O Mg+ Br2 OH CO2 MgBr(OH) Benzoic acid
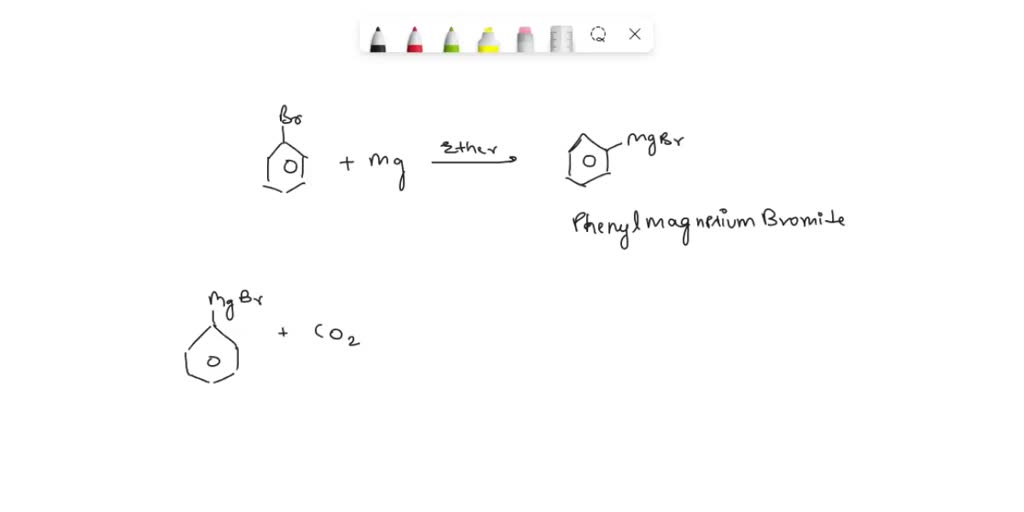
SOLVED: Ether MgBr Mg Bromobenzene Phenylmagnesium bromide MgBr ether H2O Mg+ Br2 OH CO2 MgBr(OH) Benzoic acid
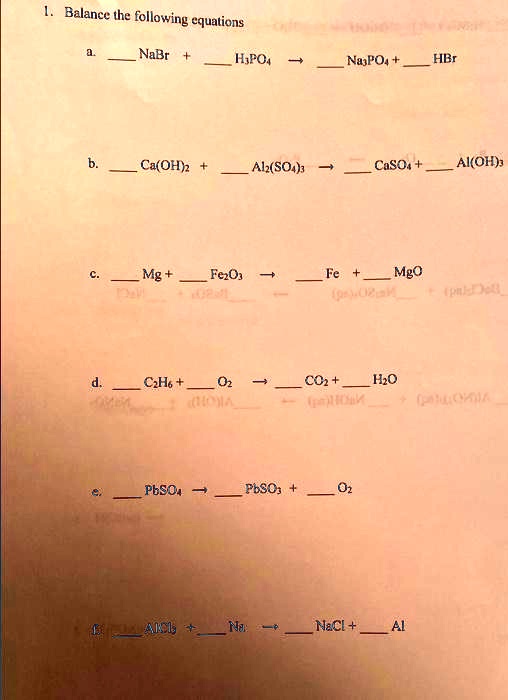
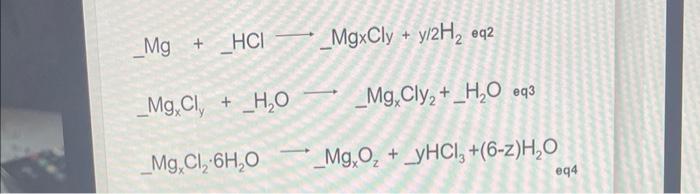
SOLVED: 1. Balance the following equations a. NaBr+ HPO4NaPO+HBr b. CaOH+ A12SO4 CaSO+ AI(OH Mg+ FeO Fe+ MgO d. CH+ CO+ H2O PbSO+ PbSO+ O N NaCl+
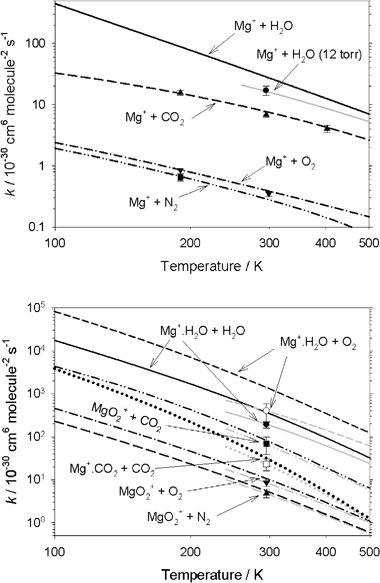
A kinetic study of Mg+ and Mg -containing ions reacting with O 3 , O 2 , N 2 , CO2 , N2O and H2O : implications for magnesium ion chemistry in

Ab Initio Studies on the Mechanism of the Size‐Dependent Hydrogen‐Loss Reaction in Mg+(H2O)n - Siu - 2002 - Chemistry – A European Journal - Wiley Online Library

Hydrated Magnesium Cations Mg+(H2O)n, n ≈ 20–60, Exhibit Chemistry of the Hydrated Electron in Reactions with O2 and CO2 | The Journal of Physical Chemistry A

Hydrated Magnesium Cations Mg+(H2O)n, n ≈ 20–60, Exhibit Chemistry of the Hydrated Electron in Reactions with O2 and CO2 | The Journal of Physical Chemistry A

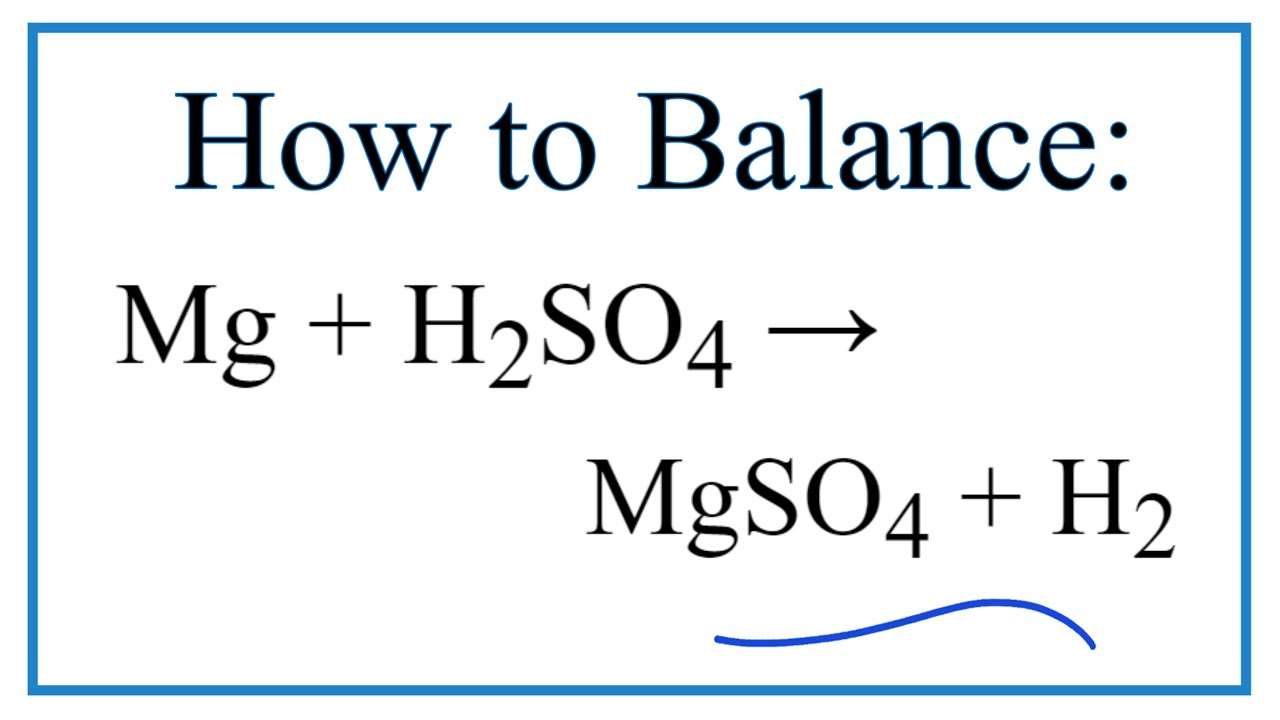
N Ctrl Alt 14. Zn + H,SO4--->ZnSO4 +S+H20 15. Mg+ H2 SO4 16. Fe+ HNO3 17. Al+ H2SO4 --->Al2(SO4)3 +H2S+ H2O 18. Mg+ HNO3--> Mg(NO3)2+N2O + H2O 19.