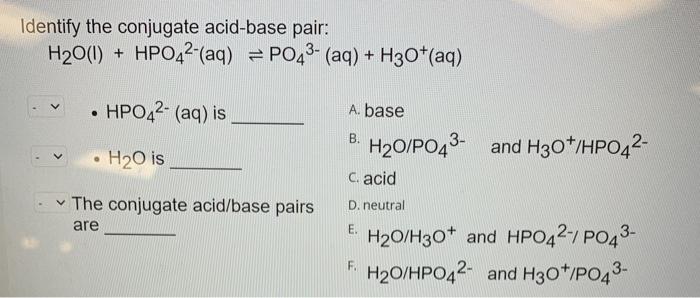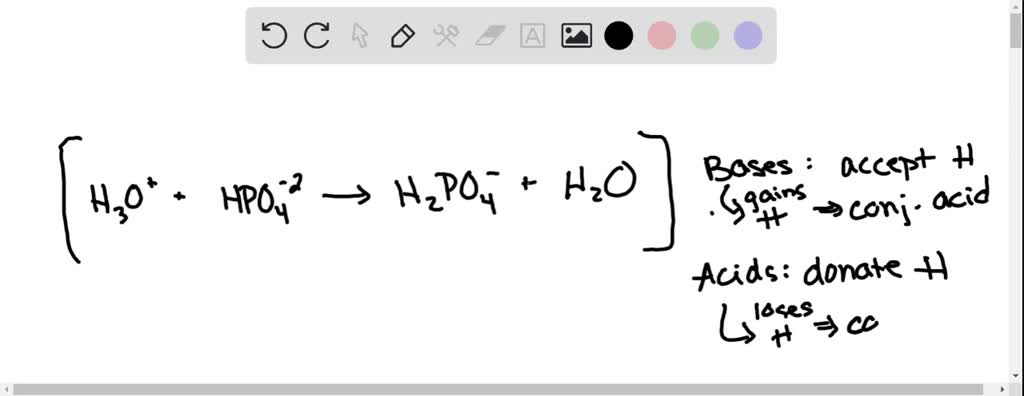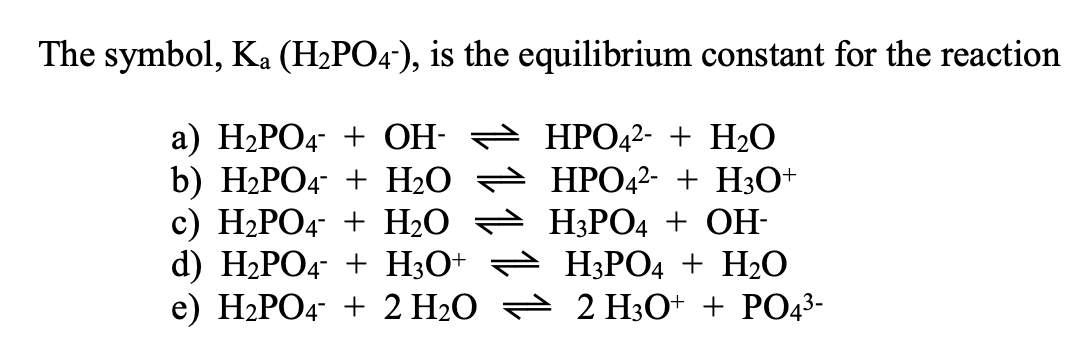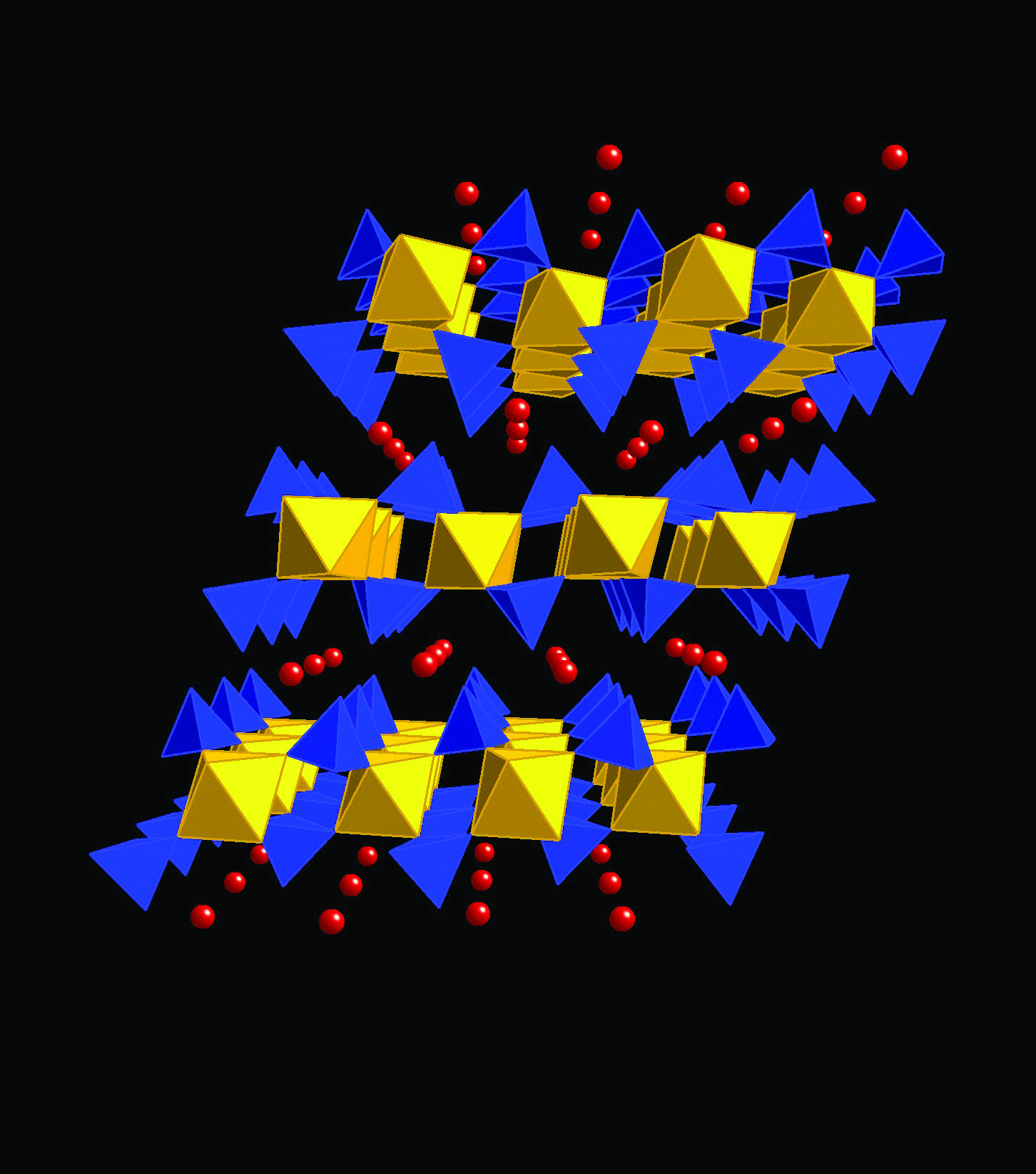
High resolution powder diffraction studies of mixed-metal layered phosphates - - Diamond Light Source
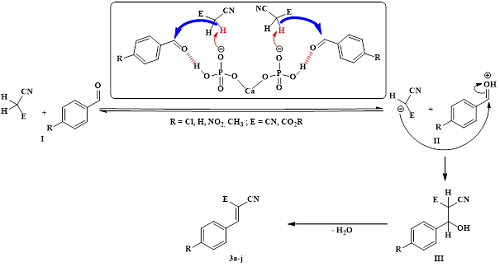
A new process for Na2Ca(HPO4)2 synthesis and its application as a heterogeneous catalyst in Knoevenagel condensation | Chehab | Mediterranean Journal of Chemistry

Catalytic Reduction of 4-Nitrophenol to 4-Aminophenol Using Ag@α-Ti(HPO4)2· H2O: Experimental and Computational Studies | Industrial & Engineering Chemistry Research

Catalytic conversions of isocyanate to urea and glucose to levulinate esters over mesoporous α-Ti(HPO4)2·H2O in green media - X-MOL

PDF) Neutron powder diffraction study of α-Ti(HPO4)2.H2O and α-Hf(HPO4)2.H2O; H-atom positions | Pilar Pertierra - Academia.edu

SOLVED: B. Identify the conjugate acid-base pairs in the following reactions: H2PO4- + OH- → HPO4-2 + H2O HBr + H2O → H3O+ + Br- CO3-2 + H2O → HCO3- + OH-
Synthesis and structural characterisation of solid titanium(IV) phosphate materials by means of X-ray absorption and NMR spectro

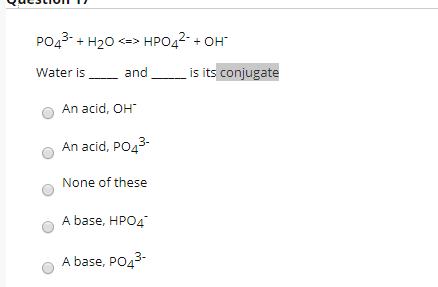
SOLVED: The following reaction is taking place in aqueous solution: HPO42- + H2O -> H3O+ + PO43-. Which is a Bronsted-Lowry base in this reaction? Select one: a. H+ b. H2PO4- c.

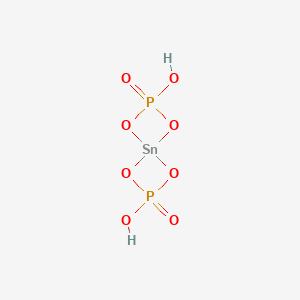
PDF) Thermal decomposition kinetics and reversible hydration study of the Li2Zn(HPO4)2�H2O | Chanaiporn Danvirutai - Academia.edu
![Na2[(VO)2(HPO4)2(C2O4)]·2H2O: A Promising Mixed Polyanionic Cathode Material for Aqueous Zn-Ion Batteries Na2[(VO)2(HPO4)2(C2O4)]·2H2O: A Promising Mixed Polyanionic Cathode Material for Aqueous Zn-Ion Batteries](https://pubs.acs.org/cms/10.1021/acs.inorgchem.2c03308/asset/images/medium/ic2c03308_0004.gif)
Na2[(VO)2(HPO4)2(C2O4)]·2H2O: A Promising Mixed Polyanionic Cathode Material for Aqueous Zn-Ion Batteries

Figure 6 from VIIIVIV(HPO4)4·enH·H2O: a mixed-valence vanadium phosphate with an open framework | Semantic Scholar