1) H2O2 + O3 → H2O +2O2 2)H2O2 +Ag2O →2Ag +H2O +O2 Determine whether H2O2 is oxidised or reduced in the above reaction? Explain.
Why is the answer B? Can someone explain this to me and why other options are incorrect. I assumed that H2O2 will decompose rapidly to form H20 and O2 with MnO2 as

Section B Enthalpy of formation & Enthalpy of combustion and Bomb calorimeter The enthalpy of the reaction H202(0)+ H2O(+ 1/2O2(g) is - 23.5 kcal mol and the enthalpy of fornuation of 120 ()

4 (i) H2O2 + 03--> H2O + 202 (ii) H202+ Ag20 >2Ag + H2O+O2 Role of hydrogen peroxide in the above reaction is respectively: (1) Oxidizing in (i) and reducing in (i) (

2H202 alkaline medium *2H20 + 02 the proposed mechanism is as given below : (1) H2O2 +1 → H2O+IO (slow) (2) H202 + 10 + H20+1+02 (fast) (i) Write rate law the
I) H2O2 + O3 → H2O + 2O2 (II) H2O2 + Ag2O → 2Ag + H2O + O2 Role of hydrogen peroxide in the - Sarthaks eConnect | Largest Online Education Community
Free Online Help: Given the following delta H values H2+1/2O2--->H2O delta H =-285.8 H2O2---->H2+O2 delta H = 187.6 Calculate delta H rxn for the following reaction H2O2--->H2O + 1/2O2

Comment équilibrer : H2O2 → O2 + H2O (peroxyde d'hydrogène, dioxygène, eau) | Physique-Chimie - YouTube

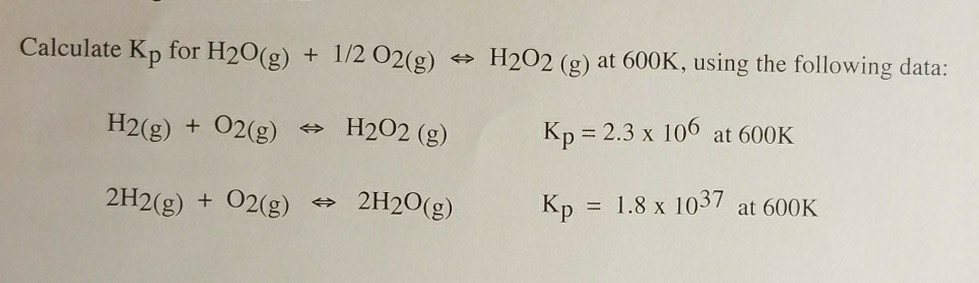
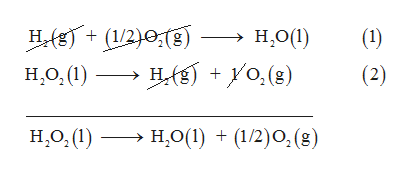












![Odia] H2O2 to H2O + 1/2 O2 is order reaction. Odia] H2O2 to H2O + 1/2 O2 is order reaction.](https://static.doubtnut.com/ss/web-overlay-thumb/11729098.webp)